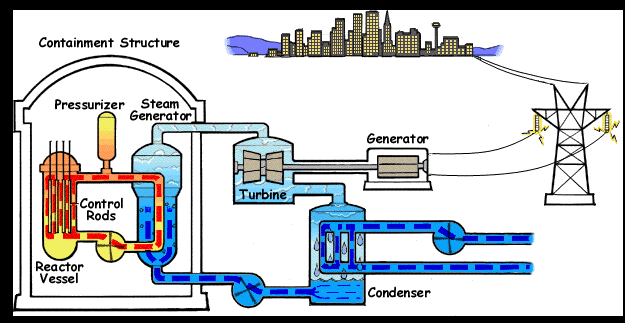
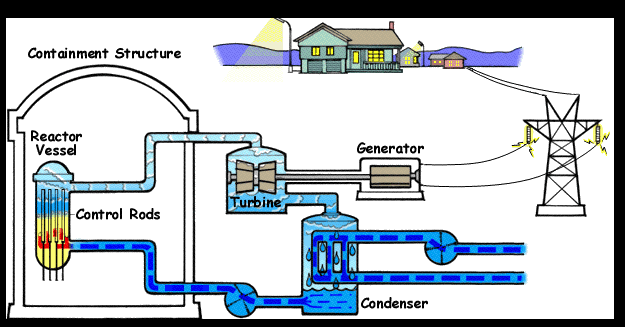
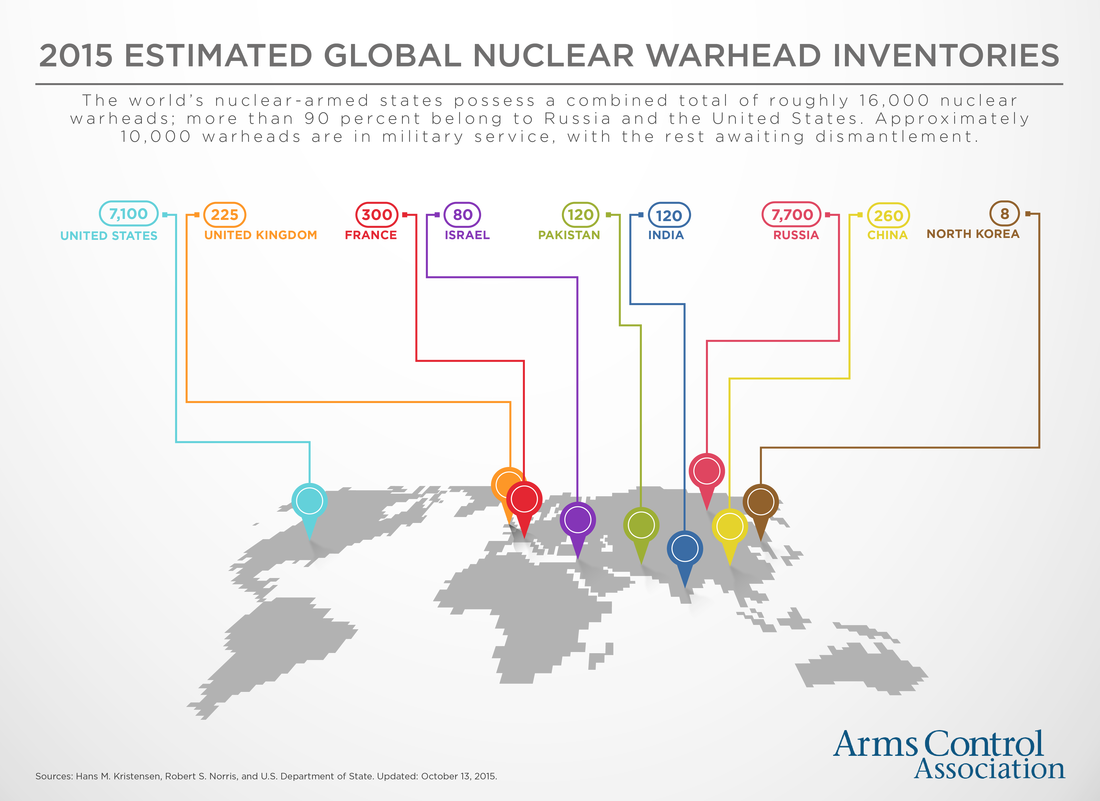
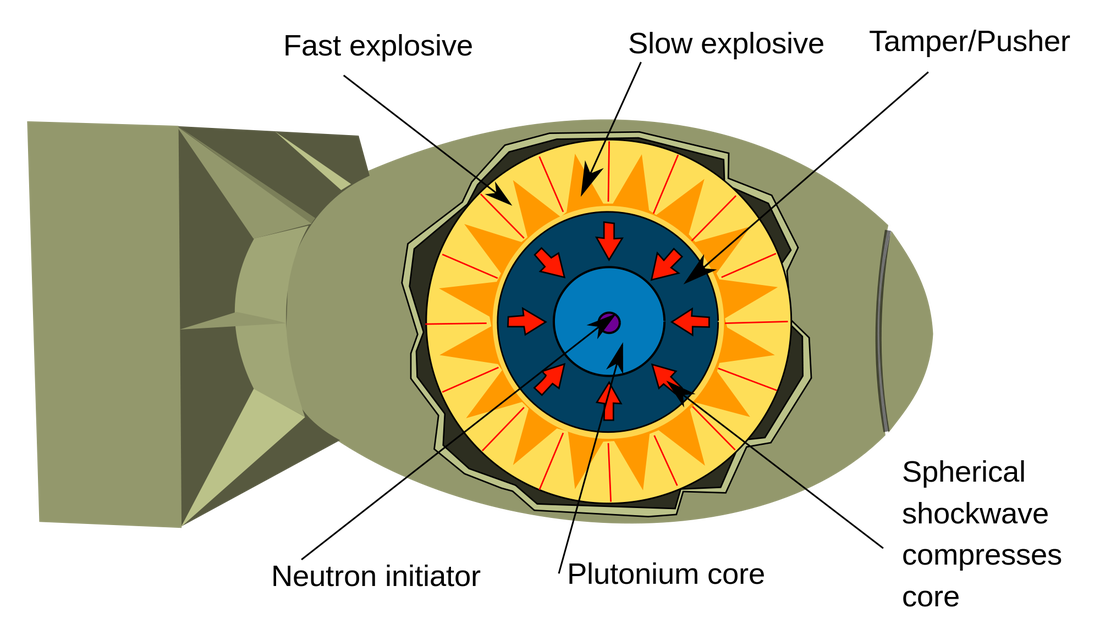
- Name, Symbol – Uranium, U
- Atomic Number: 92
- Atomic Weight: 238.02891
- Phase at Room Temperature: Solid
- Melting Point: 1135°C
- Boiling Point: 4131°C
- Electron configuration: [Rn] 5f3 6d1 7s2
- Heavy metal
- Used as an abundant source of concentrated energy
The Uranium Atom
On a scale arranged according to the increasing mass of their nuclei, uranium is one of the heaviest of all the naturally-occurring elements (Hydrogen is the lightest). Uranium is 18.7 times as dense as water. Like other elements, uranium occurs in several slightly differing forms known as 'isotopes'. These isotopes differ from each other in the number of uncharged particles (neutrons) in the nucleus. Natural uranium as found in the Earth's crust is a mixture largely of two isotopes: uranium-238 (U-238), accounting for 99.3% and uranium-235 (U-235) about 0.7%. The isotope U-235 is important because under certain conditions it can readily be split, yielding a lot of energy. It is therefore said to be 'fissile' and we use the expression 'nuclear fission'.
History
Uranium was discovered in 1789 by Martin Klaproth, a German chemist, in the mineral called pitchblende. While he was working in his experimental laboratory in Berlin in 1789, Klaproth was able to precipitate a yellow compound by dissolving pitchblend e in nitric acid (HNO3) and neutralizing the solution with sodium hydroxide (NaOH). Klaproth assumed the yellow substance was the oxide of a yet-undiscovered element and heated it with charcoal to obtain a black powder, which he thought was the newly discovered metal itself (in fact, that powder was an oxide of uranium). He named the newly discovered element after the planet Uranus, named after the primordial Greek god of the sky, which had been discovered eight years earlier by William Herschel.
In 1841, Eugène-Melchior Péligot, Professor of Analytical Chemistry, isolated the first sample of uranium metal by heating uranium tetrachloride with potassium.
Henri Becquerel discovered radioactivity by using uranium in 1896. Becquerel made the discovery in Paris by leaving a sample of a uranium salt, K2UO2(SO4)2 (potassium uranyl sulfate), on top of an unexposed photographic plate in a drawer and noting that the plate had become "fogged". He determined that a form of invisible light or rays emitted by uranium had exposed the plate.
According to the theories developed, the Earth's uranium was produced in one or more supernovae ("An explosive brightening of a star in which the energy radiated by it increases by a factor of ten billion ... A supernova explosion occurs when a star has burned up all its available nuclear fuel and the core collapses catastrophically." - Oxford Dictionary of Physics).
So, we know that the Earth's uranium was produced through this process in one or more supernovae, and that this material was inherited by the solar system of which the Earth is a part.
Where does Uranium occur?
Uranium can be found in soils and waters due to the breakdown (weathering) of rocks containing it. Once it is in the soil and water, it can be taken up by plants and consumed by people or grazing animals, or it can dissolve in the water to be consumed by any organism. Uranium deposits occur in many different rock types from sedimentary to volcanic.
- Atomic Number: 92
- Atomic Weight: 238.02891
- Phase at Room Temperature: Solid
- Melting Point: 1135°C
- Boiling Point: 4131°C
- Electron configuration: [Rn] 5f3 6d1 7s2
- Heavy metal
- Used as an abundant source of concentrated energy
The Uranium Atom
On a scale arranged according to the increasing mass of their nuclei, uranium is one of the heaviest of all the naturally-occurring elements (Hydrogen is the lightest). Uranium is 18.7 times as dense as water. Like other elements, uranium occurs in several slightly differing forms known as 'isotopes'. These isotopes differ from each other in the number of uncharged particles (neutrons) in the nucleus. Natural uranium as found in the Earth's crust is a mixture largely of two isotopes: uranium-238 (U-238), accounting for 99.3% and uranium-235 (U-235) about 0.7%. The isotope U-235 is important because under certain conditions it can readily be split, yielding a lot of energy. It is therefore said to be 'fissile' and we use the expression 'nuclear fission'.
History
Uranium was discovered in 1789 by Martin Klaproth, a German chemist, in the mineral called pitchblende. While he was working in his experimental laboratory in Berlin in 1789, Klaproth was able to precipitate a yellow compound by dissolving pitchblend e in nitric acid (HNO3) and neutralizing the solution with sodium hydroxide (NaOH). Klaproth assumed the yellow substance was the oxide of a yet-undiscovered element and heated it with charcoal to obtain a black powder, which he thought was the newly discovered metal itself (in fact, that powder was an oxide of uranium). He named the newly discovered element after the planet Uranus, named after the primordial Greek god of the sky, which had been discovered eight years earlier by William Herschel.
In 1841, Eugène-Melchior Péligot, Professor of Analytical Chemistry, isolated the first sample of uranium metal by heating uranium tetrachloride with potassium.
Henri Becquerel discovered radioactivity by using uranium in 1896. Becquerel made the discovery in Paris by leaving a sample of a uranium salt, K2UO2(SO4)2 (potassium uranyl sulfate), on top of an unexposed photographic plate in a drawer and noting that the plate had become "fogged". He determined that a form of invisible light or rays emitted by uranium had exposed the plate.
According to the theories developed, the Earth's uranium was produced in one or more supernovae ("An explosive brightening of a star in which the energy radiated by it increases by a factor of ten billion ... A supernova explosion occurs when a star has burned up all its available nuclear fuel and the core collapses catastrophically." - Oxford Dictionary of Physics).
So, we know that the Earth's uranium was produced through this process in one or more supernovae, and that this material was inherited by the solar system of which the Earth is a part.
Where does Uranium occur?
Uranium can be found in soils and waters due to the breakdown (weathering) of rocks containing it. Once it is in the soil and water, it can be taken up by plants and consumed by people or grazing animals, or it can dissolve in the water to be consumed by any organism. Uranium deposits occur in many different rock types from sedimentary to volcanic.